General obligations for health institutions when using medical devices
Published:
Changes
Health institutions is an organisation the primary purpose of which is the care or treatment of patients or the promotion of public health.
Page contents
Identification within the supply chain
Health institutions shall store and keep preferably by electronic means the UDI of the devices which they have supplied or with which they have been supplied, if those devices belong to class III implantable devices.
It is important that health institutions establish good systems and routines for traceability of all types of medical devices to make advantage of the opportunities of the UDI-system.
For further details, see MDR article 27 point 9.
«In-house» manufactured medical devices/ the exception
The regulations allow health institutions to produce, modify and use medical devices used only within health institutions in order to meet specific patient group’s specific needs, where such needs cannot be met at an appropriate level of performance by an equivalent device available on the market. Specific conditions are set for this type of manufacturing of medical devices.
For further details see MDR article 5 and IVDR article 5
Read more about in-house-manufactured medical devices (in Norwegian)
Reprocessing of single-use devices not permitted
From 1th January 2022 reprocessing of single-use devices in Norway is not permitted.
Read more about reprocessing here (in norwegian)
Conduct of a clinical investigation/performance study
Health institutions that sponsor or health professionals who participate in a clinical investigation or performance study, must be aware of their obligations.
For further details, see MDR article 72 Conduct of a clinical investigation and IVDR article 68 Conduct of a performance study.
Read more:
- Clinical investigations of medical devices
- Performance studies of in vitro diagnostic medical devices
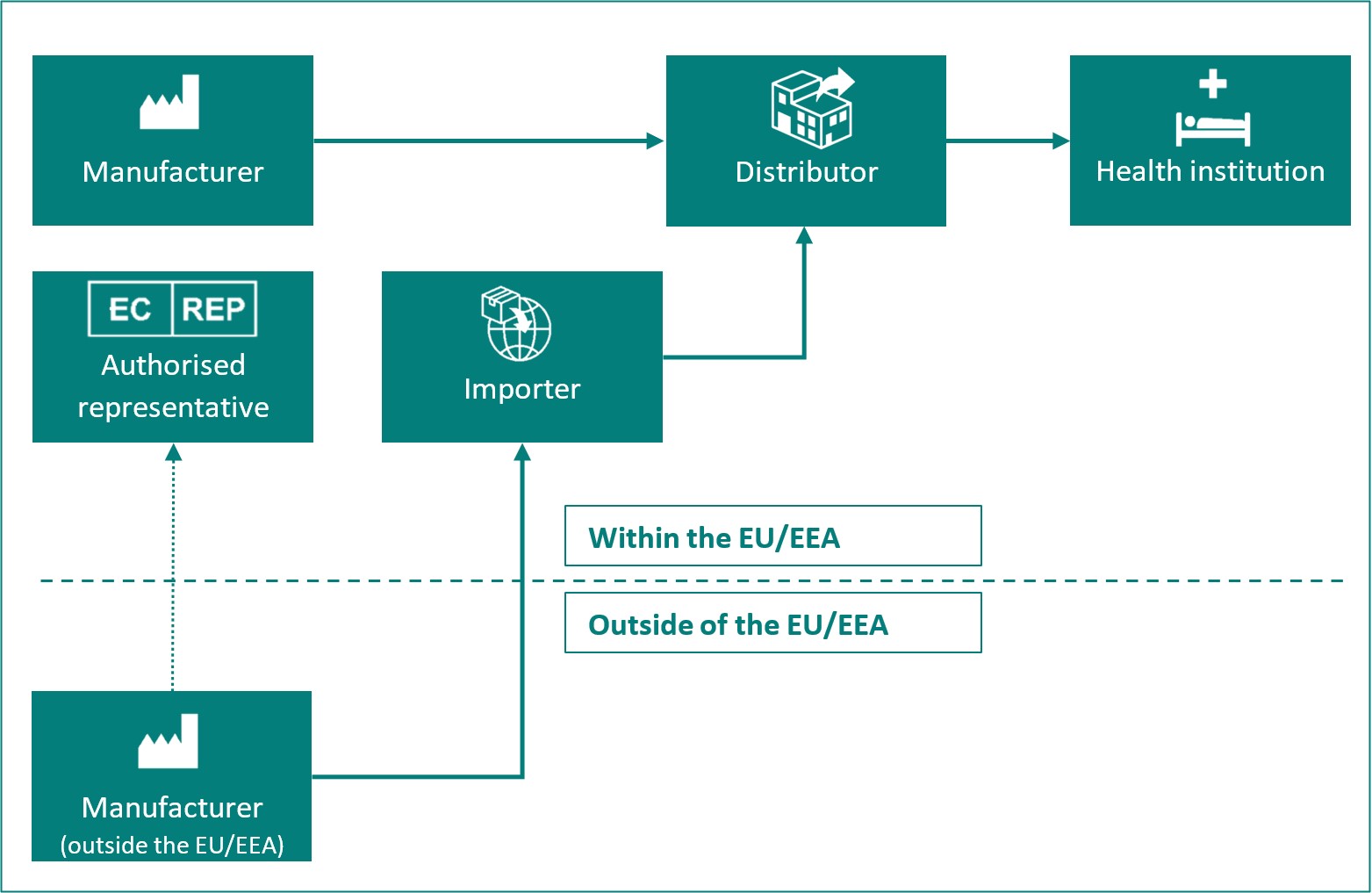
Overview of the different roles of economic operators:
| Economic operator |
Definition in MDR Article 2 and IVDR Article 2 |
| Manufacturer | A natural or legal person who manufactures or fully refurbishes a device or has a device designed, manufactured or fully refurbished, and markets that device under its name or trademark. |
| Authorised representative | Any natural or legal person established within the Union* who has received and accepted a written mandate from a manufacturer, located outside the Union*, to act on the manufacturer's behalf in relation to specified tasks with regard to the latter's obligations under this Regulation. |
| Importer | Any natural or legal person established within the Union* that places a device from a third country on the Union* market. |
| Distributor | Any natural or legal person in the supply chain, other than the manufacturer or the importer, that makes a device available on the market, up until the point of putting into service. |
| Health institution | An organisation the primary purpose of which is the care or treatment of patients or the promotion of public health. |