Parts and components that change performance or intended purpose
Published:
Changes
Changing parts and components can alter the characteristics of medical devices. Entities performing these actions have obligations.
Obligations and responsibilities
Obligations and responsibilities that economic operators assume when repairing and reprocessing a medical device that is defective or has impaired performance due to use:
-
They must ensure that the safety characteristics, performance or intended purpose of the device does not change
-
They must ensure that the actions are carried out in accordance with the manufacturer's guidelines
-
They must document that the safety characteristics, performance and intended purpose of the device have not changed
In cases where a new part or component changes the performance, safety characteristics or intended purpose of the device, it is considered a medical device and shall meet the requirements of a medical device.
In cases where repairs are made to a device intended for single use, this measure is regarded as reprocessing and entails full manufacturer responsibility. From 1 January 2022, reprocessing of single-use medical devices are not permitted.
The regulation distinguishes between repair and fully refurbishing of medical devices.
Fully refurbishing means the complete rebuilding of a device already placed on the market or put into service, or the making of a new device from used devices, to bring it into conformity with this Regulation. Fully refurbishing results in the assignment of a new lifetime to the refurbished device. Note that fully refurbishing entails full manufacturer responsibility.
Supporting documentation must be available to the Norwegian Medical Products Agency as the competent authority.
For a complete overview, see MDR Article 23 and IVDR Article 20.
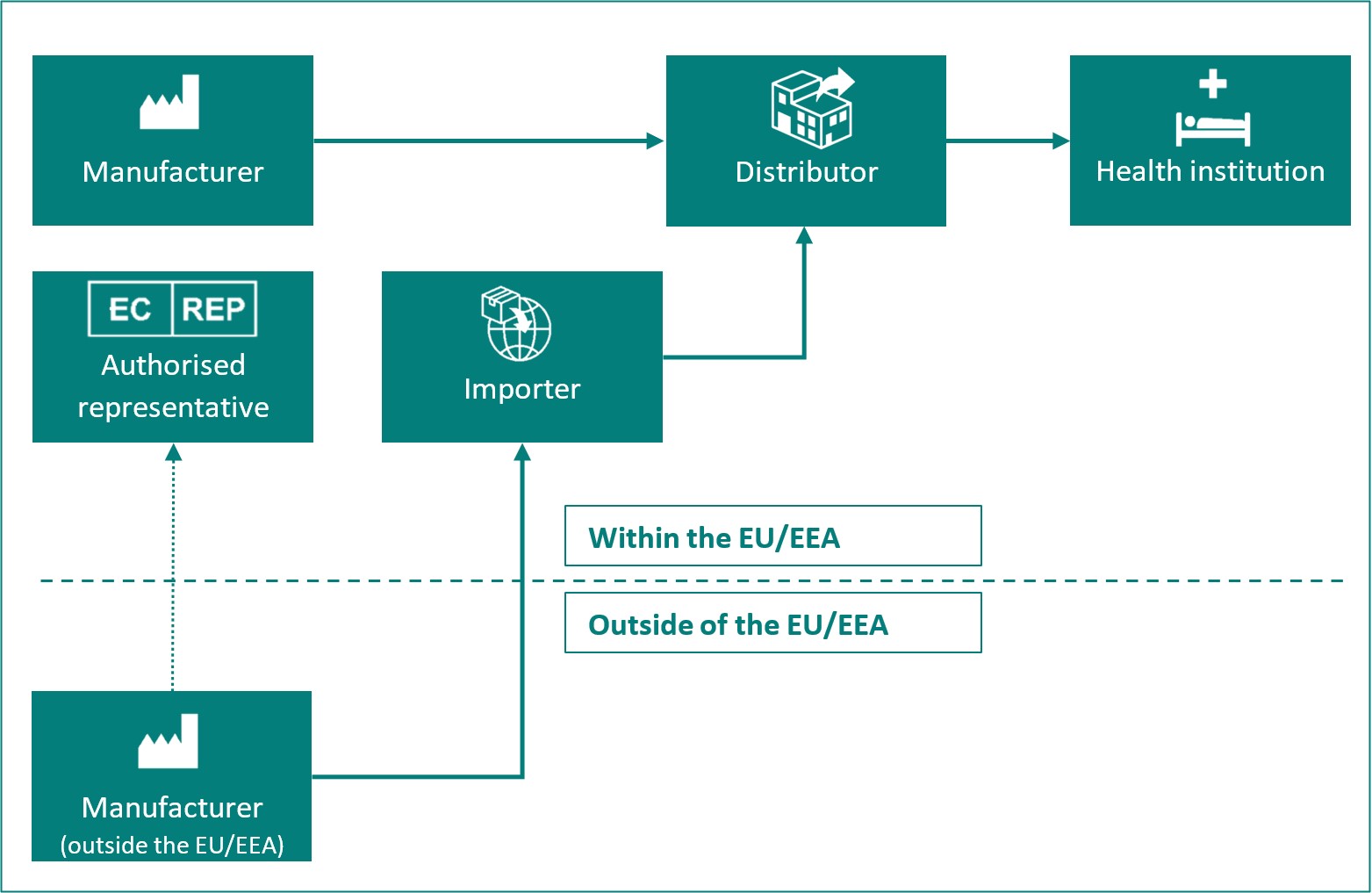
Overview of the different roles of economic operators:
| Economic operator |
Definition in MDR Article 2 and IVDR Article 2 |
| Manufacturer | A natural or legal person who manufactures or fully refurbishes a device or has a device designed, manufactured or fully refurbished, and markets that device under its name or trademark. |
| Authorised representative | Any natural or legal person established within the Union* who has received and accepted a written mandate from a manufacturer, located outside the Union*, to act on the manufacturer's behalf in relation to specified tasks with regard to the latter's obligations under this Regulation. |
| Importer | Any natural or legal person established within the Union* that places a device from a third country on the Union* market. |
| Distributor | Any natural or legal person in the supply chain, other than the manufacturer or the importer, that makes a device available on the market, up until the point of putting into service. |
| Health institution | An organisation the primary purpose of which is the care or treatment of patients or the promotion of public health. |