Field Safety Corrective Actions for healthcare facilities
Published:
Changes
Healthcare facilities are users/owners of medical devices and must receive notifications from manufacturers if a field safety corrective action has been initiated on a medical device purchased and used by the facility.
Page contents
What is a Field Safety Corrective Action?
A field safety corrective action is an action taken by the manufacturer to prevent or reduce the risk of serious incidents when using a medical device. Measures can include recalling medical device from the market, putting the device in quarantine until a solution becomes available, or making changes to the user manual/packaging leaflet accompanying the device.
What is a Field Safety Notification?
A field safety notification is a message used by the manufacturer to alert users and owners (facilities) of an affected medical device about a safety corrective action. The notification should specify the nature of the action and provide instructions on how users/owners should respond to the notification and what actions they need to perform. The person receiving the notification on behalf of the facility is responsible for communicating the safety notification to all relevant parties.
The field safety notification should clearly identify the manufacturer and the affected medical device, e.g., with relevant UDI (Unique Device Identifier). The notification should describe the cause of the field safety corrective action, the medical device malfunction, and the risks that patients, users, or others might face when using the affected device.
Field Safety Notifications in healthcare facilities
The field safety notification is considered part of the medical device instructions , similar to a user manual, and should be followed accordingly. The facility is responsible for evaluating how the field safety notification affects the healthcare provided within the facility and for ensuring compliance with healthcare legislation.
Checklist for the receipt of a Field Safety Notification
-
Carefully read the content of the field safety notification.
-
Forward the field safety notification to relevant parties.
-
Implement the measure as described in the field safety notification.
-
Provide feedback to the manufacturer if required, enabling them to monitor the corrective action.
All field safety corrective actions implemented in Europe are reviewed and accepted by the authorities in the manufacturer's home country. The content of the field safety notification is standardized across all countries, unless national guidelines require otherwise. The Norwegian Medical Products Agency evaluates all field safety corrective actions implemented in Norway.
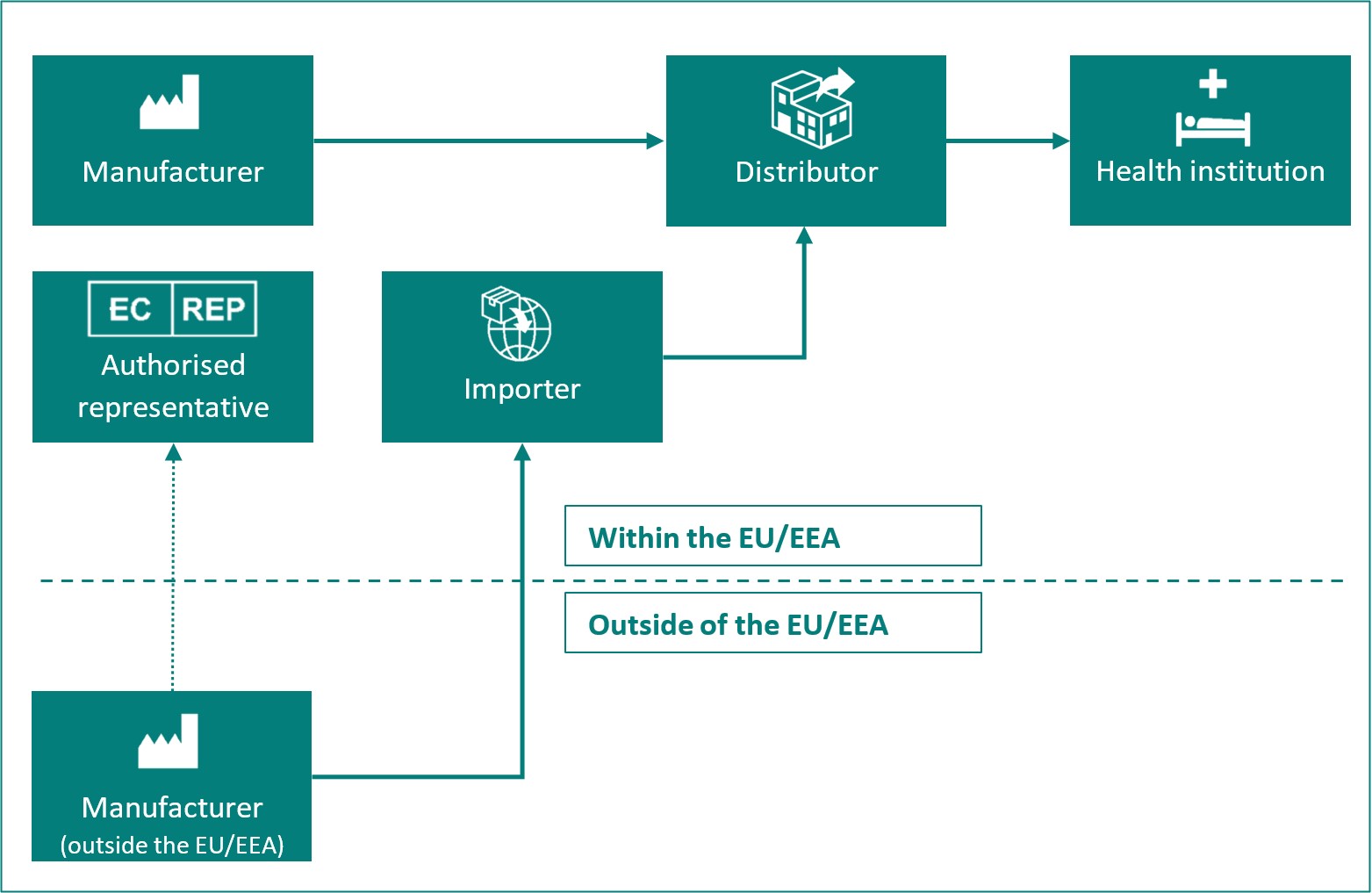
Overview of the different roles of economic operators:
| Economic operator |
Definition in MDR Article 2 and IVDR Article 2 |
| Manufacturer | A natural or legal person who manufactures or fully refurbishes a device or has a device designed, manufactured or fully refurbished, and markets that device under its name or trademark. |
| Authorised representative | Any natural or legal person established within the Union* who has received and accepted a written mandate from a manufacturer, located outside the Union*, to act on the manufacturer's behalf in relation to specified tasks with regard to the latter's obligations under this Regulation. |
| Importer | Any natural or legal person established within the Union* that places a device from a third country on the Union* market. |
| Distributor | Any natural or legal person in the supply chain, other than the manufacturer or the importer, that makes a device available on the market, up until the point of putting into service. |
| Health institution | An organisation the primary purpose of which is the care or treatment of patients or the promotion of public health. |