Market surveillance and auditing of medical devices
Published:
Changes
The Norwegian Medical Products Agency is the competent authority for medical devices in Norway. Auditing is one of the most important tools we have to ensure that medical devices that are placed on the market in Norway are safe and secure.
The Norwegian Medical Products Agency prepares annual plans for audits of economic operators. The plans are based, among other things, on risk assessment, safety monitoring data, complaints and cooperation with other European authorities.
Inspection of economic operators
Inspection of economic operators may be planned or unannounced. The economic operator is then obliged to give the Norwegian Medical Products Agency access to, among other things, the premises, warehouse and documentation we ask to see. After an inspection, a report will be prepared. The report contains an overview of the non- compliances from legal and technical requirements in accordance with the regulations for medical devices. The economic operator is given the opportunity to comment on the report before it is finalized (adopted?). In the case of non-compliances, the economic operator must prepare an action plan for how the non-compliances are to be resolved. The Norwegian Medical Products Agency may require the economic operator to provide corrective and preventative actions of deficiencies found during audits or inspections.
Some findings and deficiencies can be of such a serious degree that we may decide to make decisions about, for example, a ban on sale, recall and destruction. We can also impose a coercive fine. Cooperating European authorities will be informed of our decisions.
For further details see MDR Article 93-100 and IVDR Article 88-95.
The Norwegian Medical Products Agency is subject to the Public Administration Act and you can appeal our decisions.
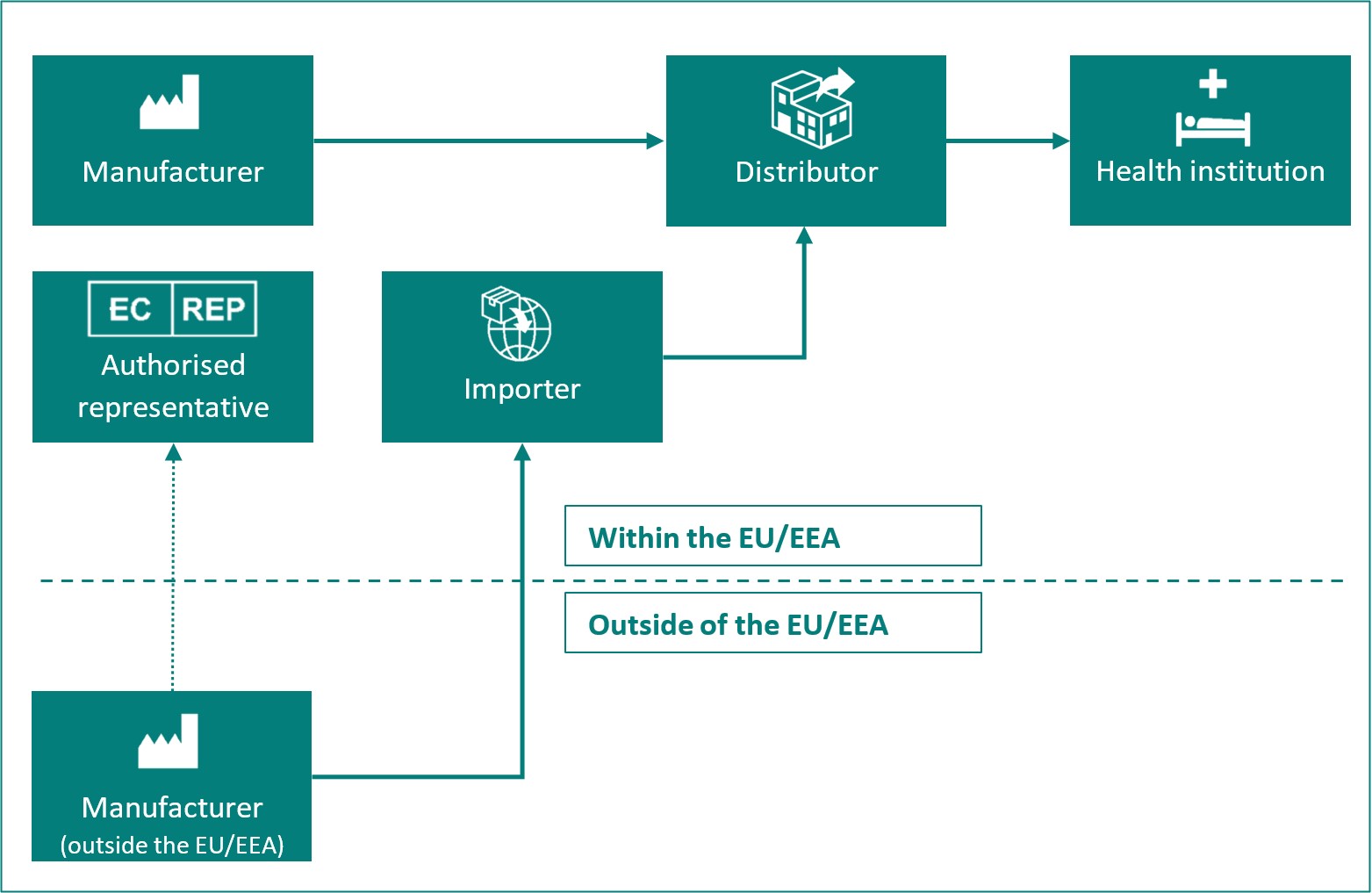
Overview of the different roles of economic operators:
| Economic operator |
Definition in MDR Article 2 and IVDR Article 2 |
| Manufacturer | A natural or legal person who manufactures or fully refurbishes a device or has a device designed, manufactured or fully refurbished, and markets that device under its name or trademark. |
| Authorised representative | Any natural or legal person established within the Union* who has received and accepted a written mandate from a manufacturer, located outside the Union*, to act on the manufacturer's behalf in relation to specified tasks with regard to the latter's obligations under this Regulation. |
| Importer | Any natural or legal person established within the Union* that places a device from a third country on the Union* market. |
| Distributor | Any natural or legal person in the supply chain, other than the manufacturer or the importer, that makes a device available on the market, up until the point of putting into service. |
| Health institution | An organisation the primary purpose of which is the care or treatment of patients or the promotion of public health. |